AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Abbott spinal cord stimulator app4/22/2024 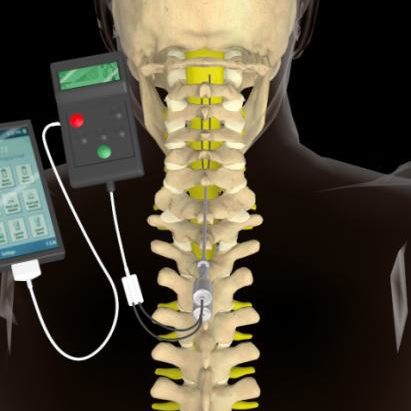
Do not perform an MRI scan on patients who have an external neurostimulation trial system or any components that are not fully implanted. The MRI scan may result in excessive heating of the system and serious patient injury. Do not perform an MRI scan on patients who have any portion of their implanted system exposed due to skin erosion. 
This can be confirmed with X-ray imaging of the neck, head, and extremity regions or by referring to the patient records. Head or extremity MRI scans can be conducted safely using a Detachable Head or Extremity RF transmit-receive coil when no parts of the implanted neurostimulation system are within the transmit-receive coil according to the conditions specified for each system in the MRI labeling. MRI scans of implants that are not located in approved locations can possibly result in increased unintended stimulation, excessive heating at the lead electrodes, and serious patient injury. Lead tips can be located at different spinal epidural levels. Two leads should travel in close proximity to one another from the IPG to the spine. The MR Conditional leads must be implanted in the epidural space and routed subcutaneously to the IPG pocket. Location of implanted system. To meet the MR Conditional requirements, components must be implanted according to the approved locations specified by the MRI labeling. MRI scans of nonfunctional leads may result in excessive heating occurring at the location of the implanted lead electrodes and serious patient injury. 
There may be a problem with the implanted lead(s)” message displays when attempting to enter MRI mode on the patient controller. Do not perform an MRI scan on patients when the “MRI is Not Advised. Do not perform an MRI scan on patients who have an incomplete neurostimulation system, where a lead is present without the IPG or disconnected from the system. Serious patient injury could occur.Ībandoned devices. Do not perform an MRI scan on patients who have any components of a neurostimulation system that are unapproved for use in an MR environment. The following warnings, precautions, and potential adverse events apply when performing MRI scans on Abbott MR Conditional SCS/DRG systems. IPG: Upper buttock, low back, flank, or abdomen IPG: upper buttock, low back, midline, flank or abdomen IPG: upper buttock, low back, midback, flank or abdomen MRI Scans with Spinal Cord Stimulation or Dorsal Root Ganglion Stimulation.See product advisories.įor expanded patient instructions on how to prepare for an MRI scan, please see the links below: Note, the Class I recall on the Proclaim™ Family of devices and the Infinity™ DBS System initiated in July 2023 does not impact a patient's eligibility for receiving an MRI scan, the conditions for the MRI eligibility, or patient safety in an MRI environment. That's why we’ve designed our devices and technologies to fit as seamlessly as possible into this critical step in your diagnostic workflow. Certain Abbott neurostimulation systems are MR Conditional with 1.5T closed bore scanners subject to conditions in the MRI Procedures Manual.īefore your patient undergoes an MRI scan:Ĭonfirm the MR Conditional components and location of the system (this information is included on the Abbott Patient ID card).Ĭonfirm that no adverse conditions to MR scanning are present.Įnsure the patient’s neurostimulation system is in MRI mode.Ĭonfirm specific scan requirements for the patient's system, intended scan location, and RF coil type. We know that MRI images are used as a tool to visualize diagnostic imaging and help provide information that may be useful to a clinician.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
